 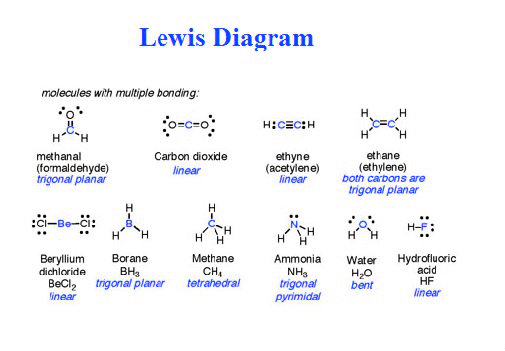
So that's it: that's the Lewis structure for B(OH)3. And the Boron only has six, but Boron is an exception and it's OK with six valence electrons. And then the Oxygens, they all have eight, so they have an octet, their shells are full. The two oxygen atoms are arranged adjacent to the central sulfur atom. We have already drawn the 2-dimensional Lewis Structure of ClF3. 
Therefore, we have got the most perfect Lewis Structure of ClF3. Sulfur is the least electronegative atom in the compound and will act as the central atom. Formal charge of each F atom Valence electrons (7) 0.5Bonding electrons (2) Lone pair of electrons ( 23) 7 1 6 0. We have Hydrogens with two valence electrons each, so their outer shells are full. Now that we’ve calculated the number of valence electrons available to us, we move on towards building up the Lewis structure for SO 2. So we've used all 24 valence electrons for the B(OH)3 Lewis structure. And then we'll fill the octets for the Oxygens: 14, 16, 18, 20, 22, and 24. We'll put two valence electrons between atoms to form chemical bonds. The way it's written suggests that we should put the Boron at the center and then the OH's around it. So 3 plus 7 times 3, that's 21 24 valence electrons for the B(OH)3 Lewis structure. Determine the total number of valence electrons. 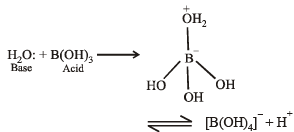
Transcript: For the B(OH)3 Lewis structure, we have 3 valence electrons for Boron and then we have 6 for Oxygen, plus 1 for the Hydrogen, but we have three of these OH's so we're going to multiply this whole thing by three. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
